Comprehensive multi-resolution investigation of IPF pathology
Dr. Dragoş M. Vasilescu, currently a Research Associate in the Department of Pathology and Laboratory Medicine, is studying pulmonary diseases such as chronic obstructive pulmonary disease (COPD) and IPF using a combination of high resolution CT, histopathological microscopy and gene-expression tools. Through an international collaboration with the lung transplant center at Katholieke University Leuven in Belgium, Dr. Vasilescu and his colleagues just published a novel study of IPF that demonstrated, for the first time, that the small airways in patients with IPF are already reduced in number in radiologically defined (on a clinical CT scan) regions of minimal fibrosis (Verleden SE et al. Lancet Respir Med. 2020 Feb 13, https://doi.org/10.1016/S2213-2600(19)30356-X).
The Parker B. Francis fellowship will enable Dr. Vasilescu to provide a better understanding of the underlying microscopic and molecular changes in IPF.
To continue this important and novel work, Dr. Vasilescu recently received 3 years of salary support from the United States-based Parker B. Francis Foundation (PBF, http://www.francisfellowships.org). PBF Fellowships are awarded to only one Canadian researcher each year; over the past 40 years, several fellows from the Centre for Heart Lung Innovation received this prestigious fellowship, all of whom later successfully transitioned to faculty positions either at UBC or at other notable universities around the world. During the course of his fellowship, Dr. Vasilescu will be mentored by Dr. James Hogg at the Centre for Heart Lung Innovation located at St. Paul’s Hospital, together with an interdisciplinary team that includes Drs. Tillie-Louise Hackett (microbiologist), Kohei Ikezoe (respirologist), Cameron Hague (radiologist), Darra Murphy (radiologist) and Chris Ryerson (respirologist).
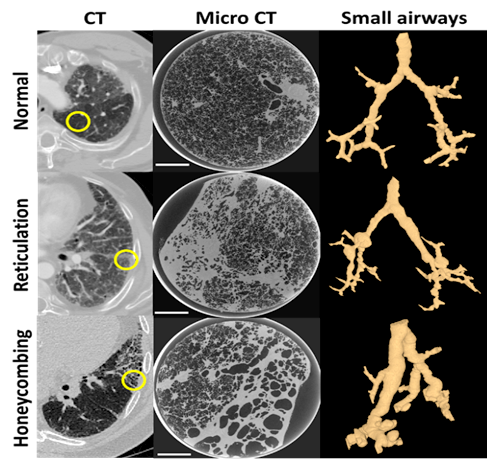
During his fellowship, Dr. Vasilescu will combine data obtained from novel tools such as micro CT, single-cell imaging mass spectrometry and next generation sequencing. The proposed study includes the following aims: Firstly, Dr. Vasilescu and his colleagues seek to understand the different types of damage that occur in IPF that can be observed using high resolution 3D imaging in the clinical setting and relate that with findings at the microscopic level using micro CT. Secondly, the team will use state-of-the-art single cell imaging to identify the cells and their gene signatures that are responsible for the distinct structural changes present in IPF. The preliminary data obtained from cryo-preserved samples from patients with IPF, scanned with a microCT scanner at 13 µm resolution, showed striking differences in the small airway morphometry in regions of radiologically-distinct patterns (Figure 1) . The novel combination of structural and molecular analysis in the above experiments will provide us with a better understanding of IPF, and the potential to improve diagnosis and find therapeutic solutions for the patients suffering from this disease.