
Dr. Jay Kizhakkedathu (centre) with the two lead co-authors Dr. Erika Siren (left) and PhD Candidate Daniel Luo (right).
"Each and every one of the organ transplant recipients each year must face this choice: do I exchange a life of organ failure for one with chronic immunosuppression?"
Organ transplantation is a life-saving therapy for those with terminal organ failure. The procedure is very intricate, but similarly, the post-operative care is crucial and just as elaborate. To reduce the incidences of rejection and organ failure, these transplant recipients often subscribe to immunosuppressant therapies. This is done to prevent the recipient’s immune system from recognizing the new organ as foreign, and for many patients this treatment course will be permanent, cumbersome, and expensive. Broad-acting immunosuppressant drugs have long been used as a blunt tool for keeping organ transplant rejection at bay. These drugs leave transplant recipients without a functioning immune system which becomes a chronic illness in itself. Each and every one of the organ transplant recipients each year must face this choice: do I exchange a life of organ failure for one with chronic immunosuppression? By bringing forward an approach that can protect the transplant without sacrificing the immune system, could bring forward new directions in transplantation and would improve the quality of life of patients.
As reported in Nature Biomedical Engineering, researchers led by Dr. Jayachandran Kizhakkedathu have developed a novel approach to immunosuppression that ultimately, seeks to eliminate the burden of chronic immunosuppression in solid organ transplant recipients. Rather than shutting down the recipient’s perfectly healthy immune system, this new method leverages potent and long-lasting localized immunosuppression. Using a non-genetic and cell-free approach, they painted the blood vessels of the organ with a protective and localized coating made up of immune suppressive glycopolymers (sugar containing polymers) that enables the transplant to both evade immune cell recognition and permanently shut down the initiation of organ rejection. The method is easily applied ex vivo by doping clinically-available organ preservation solutions with the glycopolymer among other additives during the organ preservation period.
Mouse kidneys in the modification fluid.
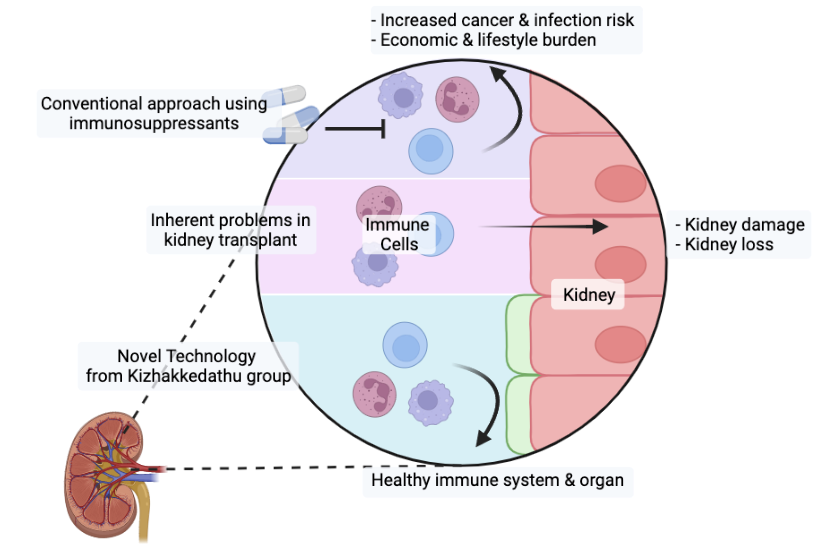
A cartoon showing comparison between classic immunosuppressants and the novel localized immunosuppression developed at the Kizhakkedathu group.
By leveraging the preservation period to swiftly append the protective layer only to the organ, the effects to suppress immune recognition and inflammation are localized and do not affect the recipient’s immune system elsewhere in the body. The mice studies illustrated promising results in hyperacute, acute and chronic organ rejection response – all in the absence of broad acting immunosuppressants. Moreover, mice transplanted with organs modified with this coating did not see hindered immunological responses elsewhere in the body, confirming its distinction from broad-acting immunosuppressive. The method has only been tested in blood vessels and kidneys in mice, but other models and organs are being investigated to assess its limitations and further confirm its translational potential to a healthcare setting. While that’s ongoing, work on new generations of this technology to further apply to various biomedical settings are also being explored (transfusion, cell transplantation and tissue engineering). Kizhakkedathu lab collaborated with Dr. Choy, at Simon Fraser University, Dr. Zhang at North Western University, and Drs. Withers, Levings and Du at UBC to accomplish their goals.